Revita Life Sciences
AVASCULAR NECROSIS OF FEMORAL HEAD (AVN)
Pathophysiology
It is because of mismatch between bone production and osteolysis with the latter occurring at a relatively faster rate. This leads to the death of the bony trabeculae supporting the articular cartilage.
Reduction of mesenchymal stem cells (MSCs) in the bone marrow while enhanced number of adipose cells and apoptotic cells is seen in AVN heads.

Core decompression (CD) is devised to release the intraosseous pressure and increase the blood flow to the femoral head, so as to prevent progression or reverse the pathology of AVN.
It has had promising results in very early stages of AVN, wherein cores of subchondral deadbone are removed by drilling, facilitating blood vessel in growth and restoration of vascularity.
CD alone in FICAT Stage 1 has shown promising results with reversal rates of up to 97% .
The success of the same procedure in FICAT Stage 2 goes down to 77%.Therefore, lies the need of adding biological to increase its efficacy.
Indication
Heads not progressed to head distortion and collapseonly up to crescent sign and <50% involvement.
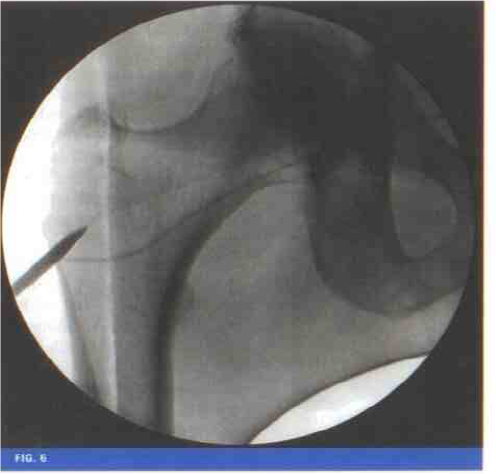
Procedure
Core decompression plus biological preferably bone marrow.30 of MNC concentrate prepared from mean volume of 180 ml aspirate
Trocar is placed under C-Arm control into the lesion
Stem cells are injected with a catheter
Hole sealed with bone plug
Results: At 2 yr
Mean volume of necrotic lesion reduced by 30 percent at 24 months
90% patients – significant improvement in Harris Hip scores (HHS>80,).
90% patients — improvement of 60% or more in their pain intensities.
The lesions shrank significantly in 60% of ARCO I and II cases
Progression of disease to Stage III in 25 % as compared to CD only (60-70%)
10 % needed THR.